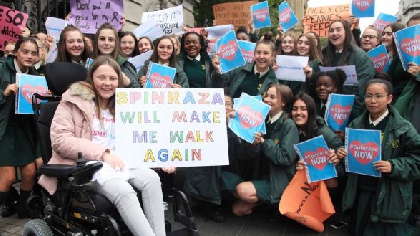
The Minister for Health, Wicklow TD, Simon Harris says he has been assured that there will be no delay in accessing the drug Spinraza, following a lengthy campaign by parents of those affected.
The HSE’s leadership team has approved access to the drug for children with type 1, 2 and 3 Spinal Muscular Atrophy on an exceptional and individualised basis.
Earlier this year, the HSE said the drug would cost about €600,000 in the first year to treat each of the 25 Irish children with the disease and €380,000 a year thereafter,.
Spinraza is now approved in 26 countries around Europe with just Estonia rejecting it.


 Wicklow Boy Awarded €45,000 for Injuries Caused by Uninsured Driver
Wicklow Boy Awarded €45,000 for Injuries Caused by Uninsured Driver
 Wicklow Woman In TG4 Documentary On Embracing Life & Defying Age Stereotypes
Wicklow Woman In TG4 Documentary On Embracing Life & Defying Age Stereotypes
 Wicklow Rents Climb As Market Tightens In Line With Rental Reforms
Wicklow Rents Climb As Market Tightens In Line With Rental Reforms
 Bray Quartet Florence Road Named Most Promising Act in Hot Press Readers’ Poll 2026
Bray Quartet Florence Road Named Most Promising Act in Hot Press Readers’ Poll 2026
 Wicklow Wide Coffee Morning Raises Almost €50,000 for Local Cancer Services
Wicklow Wide Coffee Morning Raises Almost €50,000 for Local Cancer Services



